Electrons "fill up" orbitals from the lowest energy up - that is, the second orbital does not contain an electron unless the first orbital is already full. A subshell has a place for up to two electrons. Each orbital contains one or more subshells. Here, we will only describe ground state electron configurations.Įach energy level, or shell, contains one or more orbitals. In this case, we say that the atom is in an excited state. Through energy transfers during collisions with other particles or with light, it is possible for electrons to move up to orbitals of higher energy. The farther an electron orbits from the nucleus, the higher the energy associated with it. The electrons orbit only in certain "allowed" regions around the nucleus. THE CLASSICAL MODEL OF ELECTRON ORBITAL CONFIGURATION This is a simplified scenario in which we regard the electrons as discrete objects orbiting at known, fixed distances from the nucleus. As an introduction, it's useful to present the Classical Model of atomic structure. The true nature of the electrons' movement around the nucleus is complex. The positive and negative charges cancel, and thus the net charge of the atom is zero. An atom in its neutral state always has the same number of protons as electrons. Electrons are bound to the nucleus by their electromagnetic attraction to the positively-charged protons. The electron carries a negative charge: -1e. The neutron has no charge (it is electrically neutral.) For convenience, the charges of subatomic particles are usually described as multiples of the value e, the charge of one electron, called the elementary charge.) The proton has an electric charge of +1e, or 1.602 x 10 -19 Coulombs (a Coulomb is a unit of electric charge. For the purposes of chemistry, we are interested in three of these particles: the proton, the neutron, and the electron.Īn atom consists of a central nucleus of protons and neutrons, and electrons that orbit the nucleus. Over the past two centuries, experimentalists have identified numerous particles that comprise an atom. It is not indivisible, though, as the Greek origin of its name indicates. 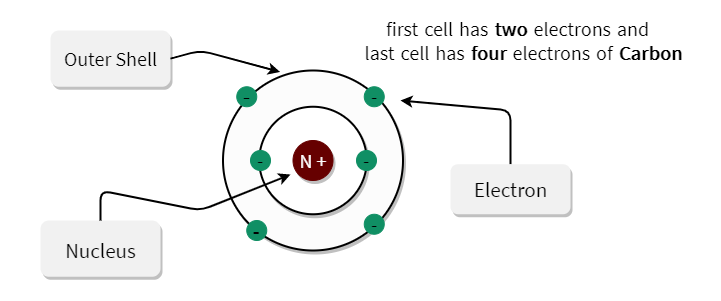
The atom is the building block of matter. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
